Webinar Date/Time: Tue, Sep 19, 2023 10:00 AM EDT
Monitoring chemical reactions and maintaining optimal conditions is critical to biochemical manufacturing. Learn more about the efficiency and effectiveness of UV-Visible absorption spectroscopy for quality control and assurance. Non Dispersive Ir Spectroscopy
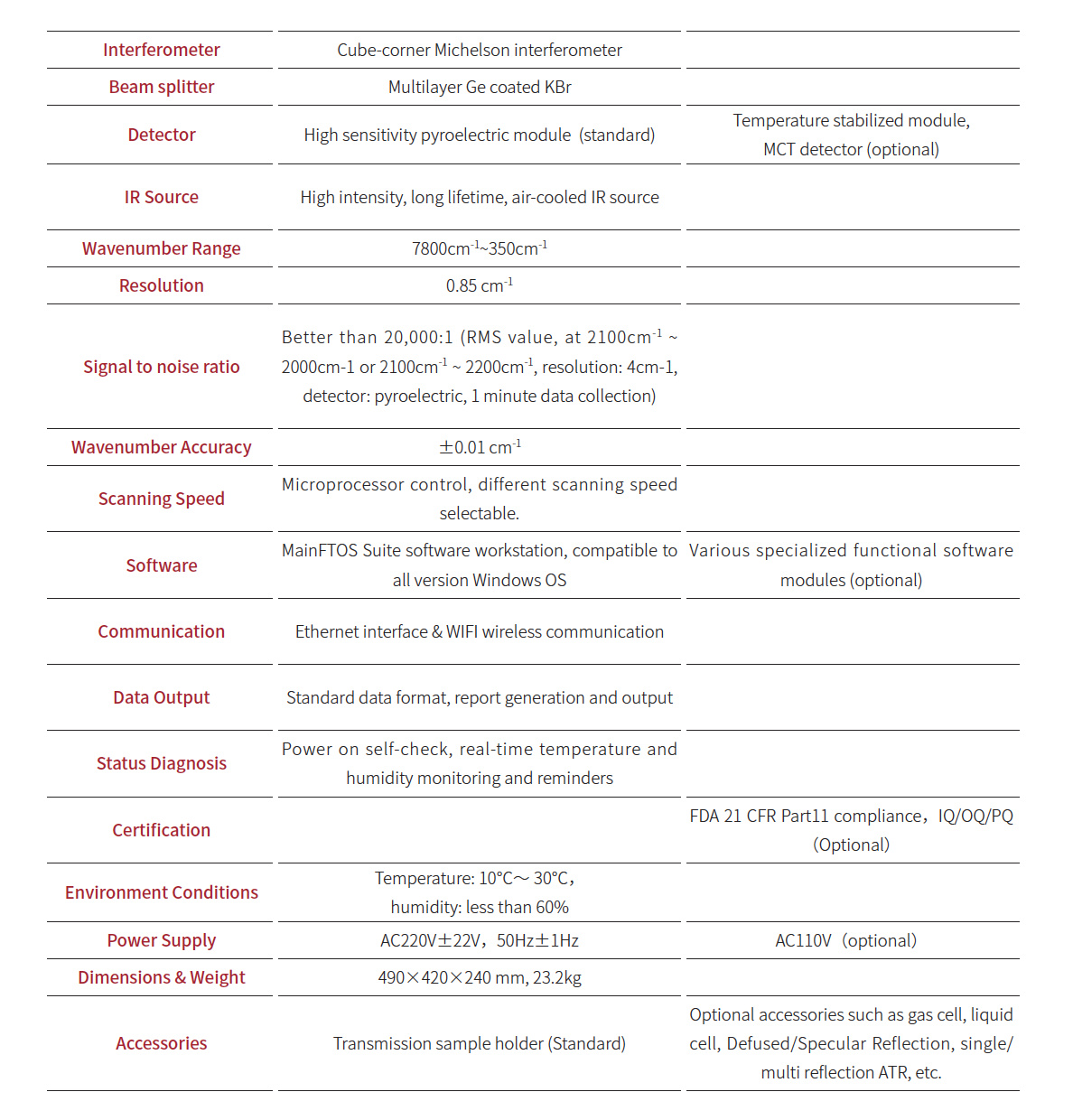
Register Free: https://www.spectroscopyonline.com/spec_w/uv-visible
From biological processes to chemical applications, synthesized materials play a key role in research and development and manufacturing across various industries. Monitoring chemical reactions during the creation of these materials is critical to optimizing output and ensuring quality. Analysis of reaction kinetics leads to understanding the overall mechanism and to ascertaining ideal reaction conditions. UV-Visible absorption spectroscopy is a productive technique for monitoring reactions. The measured absorbance is directly proportional to the concentration of a given reactant, product, or intermediate. In this webinar, hear from our expert on valuable analyses facilitated with UV-Visible measurements, as well as helpful sampling considerations for improving accuracy.
Learn how to save time, effort, and investment in biochemical manufacturing with UV-Visible spectroscopy analysis techniques.
Dr. Jennifer Empey Application Scientist Thermo Fisher Scientific
Jennifer Empey has been with Thermo Fisher Scientific as an applications scientist for over a year, supporting the cuvette-based UV-Visible instruments. Prior to this position, she received her PhD in analytical chemistry from Ohio State University where she used various spectroscopic techniques to study colloidal nanoparticles.
Register Free: https://www.spectroscopyonline.com/spec_w/uv-visible
More Than Just Acronyms at EAS 2023
Award recipient John McLean of Vanderbilt University said hybrid techniques do not exist purely as combinations of letters, slashes, and hyphens—they have been built on the shoulders of decades’ worth of analysis intended to refine and simplify workflow.
Bioprocess Monitoring with Ultrasound-Enhanced ATR Mid-IR Spectroscopy
Bernhard Lendl and Cosima Koch of the Vienna University of Technology have developed a new method for on-line monitoring of fermentations using mid-infrared spectroscopy.
Raman Spectroscopy: Recent Advancements in Pharmaceutical Analysis
An overview of Raman spectroscopy and its applications in pharmaceutical analysis is presented.
Spectroscopy and Spectroscopic Analytical Techniques in the United States Pharmacopeia–National Formulary (USP–NF)
The United States Pharmacopeia–National Formulary (USP–NF) has contributed to ensuring the quality of dietary supplements, foods, and medicines for more than 200 years. This overview explains the use of vibrational spectroscopy techniques in meeting USP–NF requirements and how the information is organized.
Process Analytical Technology (PAT) Model Lifecycle Management
The accuracy of prediction models based on spectroscopic measurements can be influenced by a variety of factors, including aging equipment. Continuous monitoring is key to managing the PAT model lifecycle and ensuring that changes over time do not negatively affect prediction model performance.
Resolving Analytical Challenges in Pharmaceutical Process Monitoring Using Multivariate Analysis Methods: Applications in Process Understanding, Control, and Improvement
In this review, we show a wide range of examples of the expanding use of multivariate analysis (MVA) in pharmaceutical manufacturing and control. MVA is being used to resolve numerous analytical challenges, such as overcoming matrix effects, extracting reliable data from dynamic matrices, and more.
More Than Just Acronyms at EAS 2023
Award recipient John McLean of Vanderbilt University said hybrid techniques do not exist purely as combinations of letters, slashes, and hyphens—they have been built on the shoulders of decades’ worth of analysis intended to refine and simplify workflow.
Bioprocess Monitoring with Ultrasound-Enhanced ATR Mid-IR Spectroscopy
Bernhard Lendl and Cosima Koch of the Vienna University of Technology have developed a new method for on-line monitoring of fermentations using mid-infrared spectroscopy.
Raman Spectroscopy: Recent Advancements in Pharmaceutical Analysis
An overview of Raman spectroscopy and its applications in pharmaceutical analysis is presented.
Spectroscopy and Spectroscopic Analytical Techniques in the United States Pharmacopeia–National Formulary (USP–NF)
The United States Pharmacopeia–National Formulary (USP–NF) has contributed to ensuring the quality of dietary supplements, foods, and medicines for more than 200 years. This overview explains the use of vibrational spectroscopy techniques in meeting USP–NF requirements and how the information is organized.
Process Analytical Technology (PAT) Model Lifecycle Management
The accuracy of prediction models based on spectroscopic measurements can be influenced by a variety of factors, including aging equipment. Continuous monitoring is key to managing the PAT model lifecycle and ensuring that changes over time do not negatively affect prediction model performance.
Resolving Analytical Challenges in Pharmaceutical Process Monitoring Using Multivariate Analysis Methods: Applications in Process Understanding, Control, and Improvement

Liquid Gas Chromatography In this review, we show a wide range of examples of the expanding use of multivariate analysis (MVA) in pharmaceutical manufacturing and control. MVA is being used to resolve numerous analytical challenges, such as overcoming matrix effects, extracting reliable data from dynamic matrices, and more.